Have you ever wondered why some atoms are like little magnets? In the world of science, atoms love to play a game of tug-of-war. They fight over tiny things called electrons. Some atoms are very strong and pull hard, while others are quite weak. This pulling power is what scientists call electronegativity. When we look at the periodic table, we see a special pattern called the electronegativity trend. This pattern helps us guess how different elements will act when they meet each other. It is like having a map for a big treasure hunt in a lab.
Understanding the Basics of Atoms
Every single thing you see is made of atoms. These tiny building blocks have a center called a nucleus and little bits called electrons spinning around them. Atoms want to be happy and stable, so they often share or steal electrons from their neighbors. The electronegativity trend tells us which atoms are the biggest “bullies” in the playground. If an atom has high electronegativity, it pulls electrons toward itself very strongly. If it has low electronegativity, it is more likely to give its electrons away to a friend.
Why the Electronegativity Trend Matters
You might ask why we need to know about the electronegativity trend in our daily lives. Well, this trend explains why water stays together and why salt tastes the way it does. When atoms bond, their pulling power decides what kind of bond they make. If the pull is equal, they share nicely. If one is much stronger, it takes the electron for itself. Knowing the electronegativity trend helps scientists create new medicines and even better batteries for your favorite toys and gadgets.
Moving Across the Periodic Table
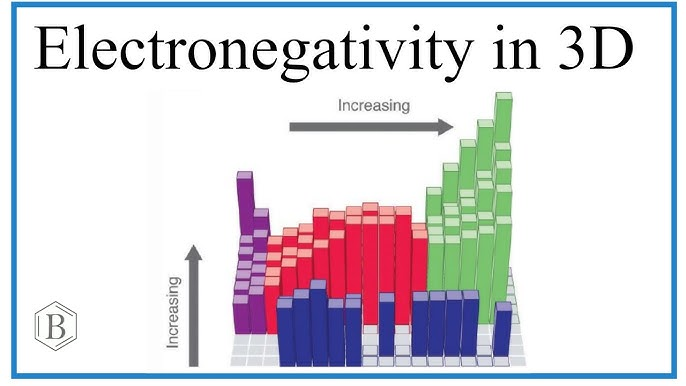
As you move from the left side to the right side of the periodic table, things change. This is a big part of the electronegativity trend. On the left, atoms are big and have fewer protons in their center. They do not pull very hard. But as you go right, the atoms get more protons. These protons act like a stronger magnet. This makes the electronegativity trend go up as you move toward the right side. It is like a staircase where each step makes the pull much stronger than the last one.
Moving Up and Down the Columns
The electronegativity trend also changes if you go up or down a column. Think of an atom like an onion with many layers. Atoms at the bottom have many layers of electrons. These layers block the pull from the center. Because of this, the electronegativity trend goes down as you move toward the bottom of the table. Atoms at the top are small and have fewer layers. This lets their center pull much harder on new electrons. This is why the top right corner is the strongest spot.
Meet Fluorine the King of Pull
In the world of chemistry, there is one element that wins every tug-of-war. That element is Fluorine. According to the electronegativity trend, Fluorine is the most powerful atom because it sits at the top right. It is very small and has a very strong center. It wants electrons more than any other element on the whole periodic table. If you see Fluorine in a science problem, you can bet it is pulling the hardest. It is the gold medal winner of the electronegativity trend competition.
The Role of the Pauling Scale
Scientists like to measure things to be very exact. A famous man named Linus Pauling created a special scale to rank atoms. We call this the Pauling Scale, and it maps out the electronegativity trend perfectly. He gave Fluorine a score of 4.0, which is the highest possible grade. Elements like Francium get a very low score, near 0.7. By using these numbers, we can see the electronegativity trend in action. It makes it easy to compare two different atoms without guessing who will win the pull.
How Atomic Size Affects the Pull
The size of an atom is a huge reason for the electronegativity trend. Think about holding a ball. If the ball is close to your body, it is hard for someone to take it. Small atoms keep their electrons very close to their center. This makes their pull feel very strong to outside electrons. Large atoms have electrons that are very far away. Because they are so far, the pull is weak. This is why small atoms usually sit at the top of the electronegativity trend charts.
Noble Gases and the Trend
You might notice some elements on the far right that do not seem to follow the rules. These are called Noble Gases. Most of the time, they do not have a value in the electronegativity trend. This is because they are already very happy and stable. They do not want to steal or share electrons with anyone else. They are like the kids who finished their lunch and do not want any of yours. However, the electronegativity trend still applies to almost every other element you will study in school.
Chemical Bonds and Pulling Power
When two atoms decide to stick together, they form a bond. The electronegativity trend decides what that bond looks like. If the atoms have a similar pull, they form a covalent bond. This means they share the electron fairly. If one atom follows the electronegativity trend and is much stronger, it might create an ionic bond. This is where the strong atom just takes the electron away. This determines if a substance will be a solid, a liquid, or a gas at room temperature.
Real World Examples of the Trend
Let’s look at water, which is $H_{2}O$. Oxygen is much higher on the electronegativity trend than Hydrogen is. Because Oxygen pulls harder, the electrons spend more time near the Oxygen. This makes one side of the water molecule a little bit negative and the other side a little bit positive. This “stickiness” is why water can flow and stay in a drop. Without the electronegativity trend, water wouldn’t act the way it does, and life would be very different for all of us.
Biography of Linus Pauling
| Detail | Information |
| Full Name | Linus Carl Pauling |
| Born | February 28, 1901 |
| Famous For | Electronegativity Scale and Chemical Bonds |
| Awards | Two Nobel Prizes (Chemistry and Peace) |
| Education | Oregon Agricultural College and Caltech |
| Big Idea | How atoms stick together using electrons |
Frequently Asked Questions
1. What is the easiest way to remember the electronegativity trend?
Just remember that atoms get “hungrier” for electrons as you go up and to the right. Think of Fluorine in the top right corner as the hungriest atom of all!
2. Does every element have electronegativity?
Most do, but the Noble Gases usually do not. They are already full and do not need to pull on any extra electrons from their neighbors.
3. Why does electronegativity decrease down a group?
It goes down because the atoms get bigger. The center of the atom is too far away from the outside to pull on electrons effectively.
4. Who discovered the electronegativity trend?
Linus Pauling is the most famous scientist who gave us a way to measure it. He created a scale that we still use in classrooms today.
5. Is electronegativity the same as electron affinity?
They are similar but not the same. Electronegativity is about pulling electrons in a bond, while electron affinity is about a single atom grabbing an electron.
6. Why is Fluorine the highest in the trend?
Fluorine is very small and has many protons for its size. This gives it a super strong “magnetic” pull that no other element can beat.
Conclusion
Understanding the electronegativity trend is like finding a secret key to chemistry. It explains how atoms behave, how they bond, and why the world around us stays together. By looking at the periodic table, you can now predict which atoms will be strong and which will be weak. Whether you are looking at a glass of water or a piece of metal, the electronegativity trend is at work. Keep exploring the world of science, and you will see these patterns everywhere! If you enjoyed learning this, why not share this guide with a friend?